Standard Potentials
This cell has a copper electrode in 1.0 M Cu(NO3)2 and an iron electrode in 1.0 M Fe(NO3)2

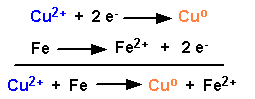
|
Standard Potentials |
|
|
This cell has a copper electrode in 1.0 M Cu(NO3)2 and an iron electrode in 1.0 M Fe(NO3)2 |
 |
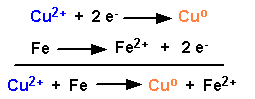 |
|
The combination of these two reactions gives 0.78 volts.
Let's assign a voltage to each reaction so we can predict the voltage from other combinations of metals.