Molecules with Two or More Double Bonds
Many molecules contain two or more double bonds and are accordingly called dienes, trienes, and so on. Some examples of their nomenclature are given in Fig. 2-27; other examples are given among the problems at the end of the chapter. The longest chain containing as many double bonds as possible is chosen as the parent, with numbering begun at the end that gives the double bonds their smaller numbers. If the double bonds are separated by at least one saturated carbon atom, as in 1,4-pentadiene, the two have little effect on one another and react independently. Bonds of this type are said to be isolated or unconjugated double bonds (Fig. 2-27). The term unconjugated double bonds also includes cumulated double bonds, as in propadiene (Fig. 2-27), but we shall have little to say about these relatively rare cases. Each double bond in a system of isolated double bonds reacts exactly as it would if the molecule contained only one double bond.
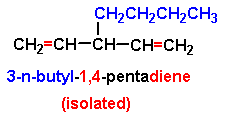


Figure 2-27. If double bonds are separated by one or more saturated carbon atoms, they are said to be isolated. If they are separated by only one single bond, they are said to be conjugated. Two double bonds on one carbon are said to be cumulated.
If the double bonds are separated by just a single bond, as in 1,3-pentadiene, they are said to be conjugated, and conjugated double bonds can sometimes modify the course of alkene reactions. As an example, consider the addition of one molar equivalent of HCl to 1,3-butadiene; not only the expected product from Markovnikov addition across the 1,2 double bond is formed, but also an unexpected product of 1,4 addition, in which the HCl adds across the ends of the conjugated double bonds and the remaining double bond moves to the middle (Fig. 2-28). Because systems of conjugated double bonds play such an important role in the chemistry of benzene and other aromatic hydrocarbons, synthetic rubber, and biosynthesis, we must examine this reaction more closely.

Figure 2-28. Addition to conjugated double bonds may take place simultaneously in the normal manner and at the ends of the system.
As with a simple double bond, addition of HCl to a conjugated diene proceeds in steps, by way of an intermediate carbon cation (Fig. 2-29), but now the cation is formed adjacent to a carbon-carbon double bond. Since a double bond is electron-rich, it can share its electrons with the adjacent electrondeficient carbon cation to an even greater extent than an alkyl group can. This electron-sharing is easy to see if we draw the molecular orbital (MO) picture for butadiene. As the proton uses the electron pair of one double bond to form a new C-H bond, electrons from the other pi orbital flow into the now emptying p orbital on C-2, so that in the intermediate cation a two-electron pi orbital is shared by three carbons and the positive charge is not localized on C-2 but is delocalized. We can obtain the same result just by using normal double and single bonds [valence-bond (VB) picture]. We note that a second structure can be written for the intermediate cation just by moving an electron pair from the C-3, C-4 double bond. No movement of atoms is involved here, just of electrons. These two structures (called resonance forms) for the cation clearly indicate that the positive charge is not localized on C-2 but is delocalized. Since C-2 and C-4 share the positive charge, it is easy to understand why the chloride ion might combine with either of them to form the two different products actually isolated in their reaction. Additional examples of this type of charge delocalization will be discussed in "Aromatinc Compounds."
Figure 2-29. Addition of an acid to the end of a conjugated diene gives an intermediate cation in which the charge is shared by both the C-2 and C-4-carbon atom, here shown in both a valence-bond (VB) and molecular-orbital (MO) picture. The VB picture shows clearly how only C-2 and C-4 (and not C-3) share the charge, while the MO picture makes the dispersal of charge by overlap of pi orbitals very obvious.
Copyright (c) 1999. All rights reserved.


